European Project adressing the safety
of blood transfusion
Work in progress
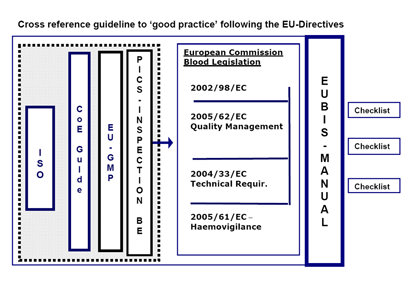
Survey results and Cross Reference Guidelines
The objective of the survey is to provide:
- an overview of the participating Blood Establishments structures and the background of inspection standards
- a definition of areas of interest and risk of the participating Blood Establishments
- a selection of the criteria commonly used for inspections
The survey was performed using a survey questionnaire distribution to the participants in this project and at competent authorities within the EU.
The survey questionnaire has been structured into 6 Sections as follows:
- Section I Activities carried out in each blood establishment
- Section II Overview of the blood establishments in each member state
- Section III Quality systems used in each blood establishment
- Section IV Inspection and audits systems used in each blood establishment
- Section V Overview of the inspection process in each blood establishment
- Section VI Objectives and deliverables of the EuBIS project
Sections I III are focused on the overall activities performed in blood establishments and should give indicators for the topics to be focused on in the inspection checklists. These sections were not completed by competent authorities. In Section II, additional information material could be enclosed.
Sections IV and V intends to provide information on the current criteria and standards used for inspections. These sections were completed by blood establishments and by competent authorities.
Evaluation of completed questionnaires
The evaluation of results is based on 36 questionnaires from Project participants and collaborating institutions covering 27 European states (24 EU members, 3 EFTA/EEA members and one state that is a candidate for joining the EU).
To allow more meaningful evaluation, the information gained from the completed questionnaires was divided into separate categories for blood establishments (BE), government authorities and competent authorities (CA).
Reference: Seidl C, Brixner V, Müller-Kuller T, Sireis W, Costello P, Cermakova Z, Delaney F, McMillan Douglas A, Nightingale M, van Galen JP , OConnell M, Siegel W, Sobaga L, de Wit J, Seifried E. Levels of quality management of blood transfusion services in Europe. Vox Sanguinis Science Series, Vol 3 (1), 2008: p54-62